Marketplace Materials

Case Study: Phase III COPD Studies
A sponsor was facing new and lingering challenges from the COVID-19 pandemic resulting in a slow & difficult patient recruitment process on two COPD trials. In order to overcome these challenges

Case Study: Phase II MS Trial
A large, global sponsor was facing increasing internal competition and needed a way to confidently expand their site network. This sponsor was also

Case Study: Phase III AAD Trial
A large, global sponsor was experiencing difficulties in hitting enrollment numbers for an Agitation in Alzheimer's Dementia trial.

Case Study: Phase III Metastatic Breast Cancer Trial
The sponsor first came to Inato with major concerns around finding enough experienced sites due to competition in the Metastatic Breast Cancer space.

Case Study: Portfolio Collaboration
A global sponsor was facing challenges in their trials brought on by the COVID-19 pandemic. With an increase of competition at large research sites and a

Case Study: Phase III MDD Trial
A large global sponsor was facing an increasingly tight timeline for a U.S. based Major Depressive Disorder trial due slow enrollment and complications with the

Overview: Inato for Sponsors
Looking for reliable site performance? Learn more about how Inato help. By connecting to Inato's verified network of community sites, sponsors can accelerate
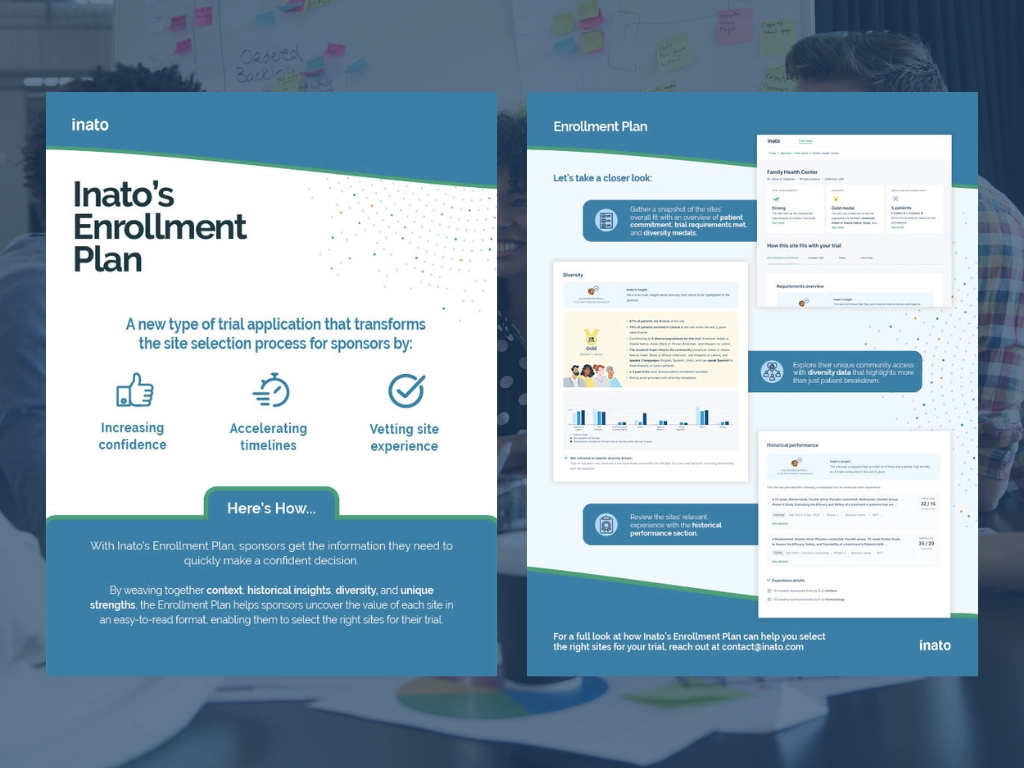
Inato's Enrollment Plan
Discover how Inato's unique trial application transforms the site selection experience and uncovers the crucial information sponsors need to select the

Inato's Inclusive Research Program
In recent history, we've made some incredible medical innovations through trials. But not everyone has access. Let's change that. Throughout the world, if you

Introducing the Inato Verified Commitment
Today, sponsors have little visibility into the process sites use to create enrollment estimates. We can fix that. For sponsors, the current process means two sites
.webp)
Overview: Inato for Sites
Explore how Inato works for sites and what other site users are saying.

Case Study: Phase III PTSD
A top global sponsor was experiencing significant delays in their trial timelines for two sister PTSD studies. Coupled with a high screen failure rate holding back