Faster trials.
Broader access.
One AI-powered platform.
Introducing your competitive advantage. Inato’s AI-powered platform connects you to engaged sites at scale, so you can accelerate drug development and ultimately bring care to the patients who need it most.




Innovating with
Forward-Thinking Pharma Leaders
We co-develop our technology with forward-thinking pharma and site leaders to ensure every trial is faster, more efficient, and more inclusive.












Why Leading Sponsors Partner with Inato
Partner with
proven sites
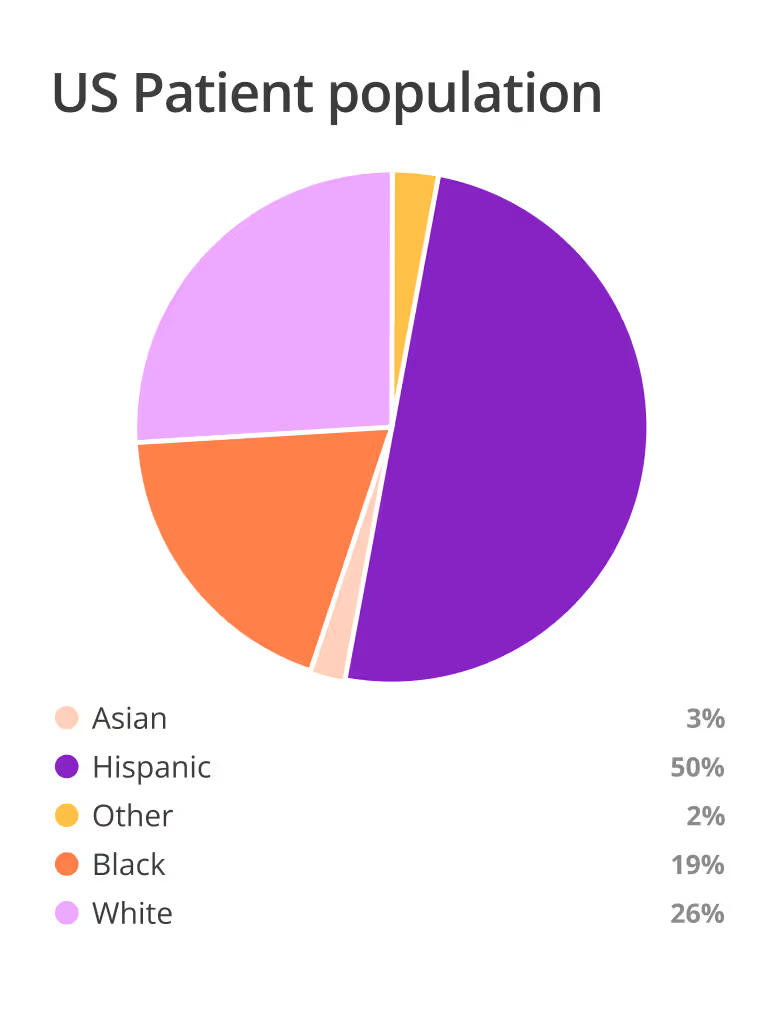
Work with motivated, high-performing sites that match your protocol and help you reach underrepresented communities.
Reach more
patients, everywhere
Connect with 6,000+ committed research sites across 50+ countries to reach the right patients, faster.
Everything in
one platform
From early planning to patient enrollment, simplify how you run trials, all in one place.
How we do it
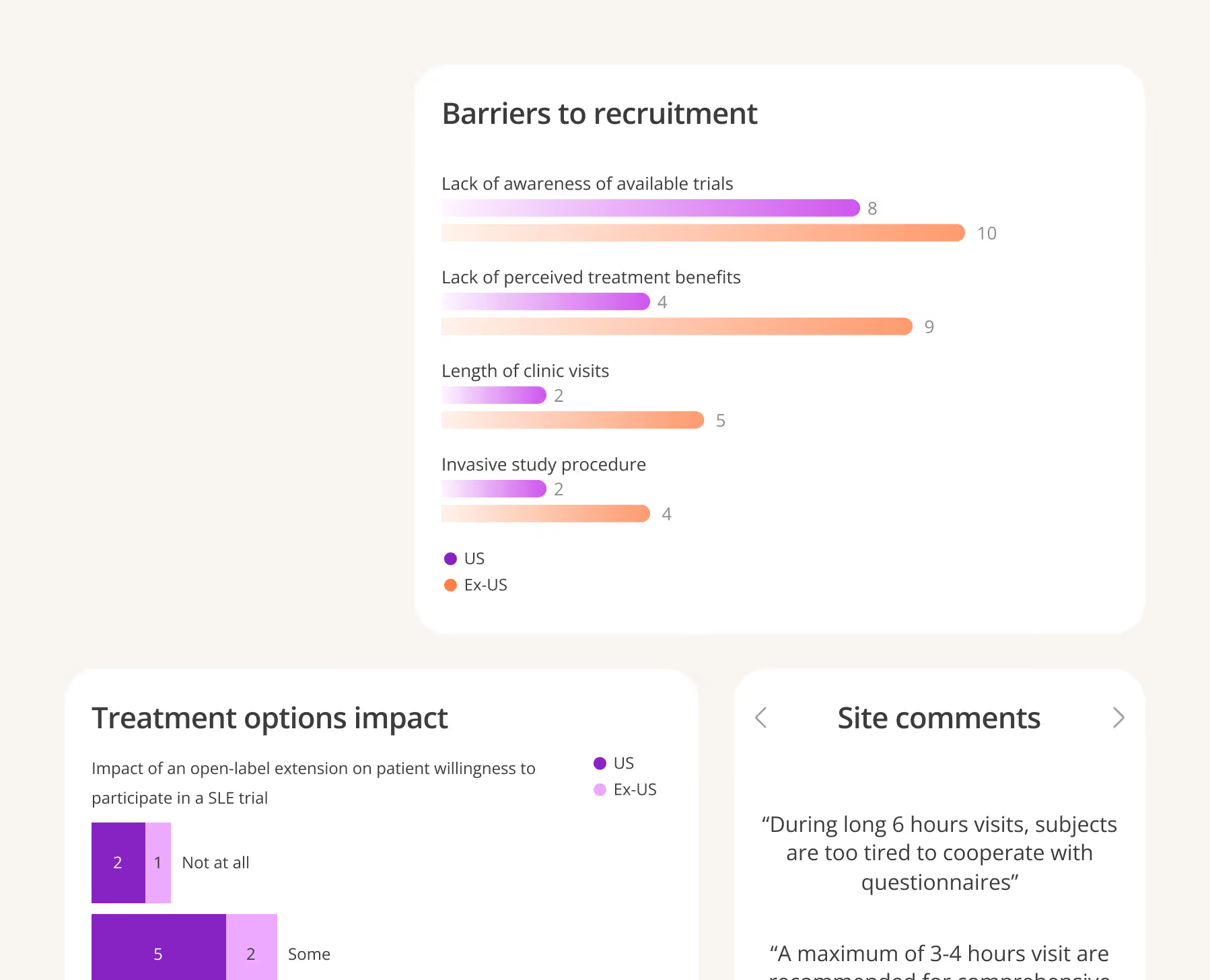
Inato’s platform is built to support you from early planning to enrollment. Make faster, more informed decisions with our co-developed AI capabilities and site insights at scale.




Early planning
Use site insights at scale to inform your strategy and design trials that are both inclusive and efficient.


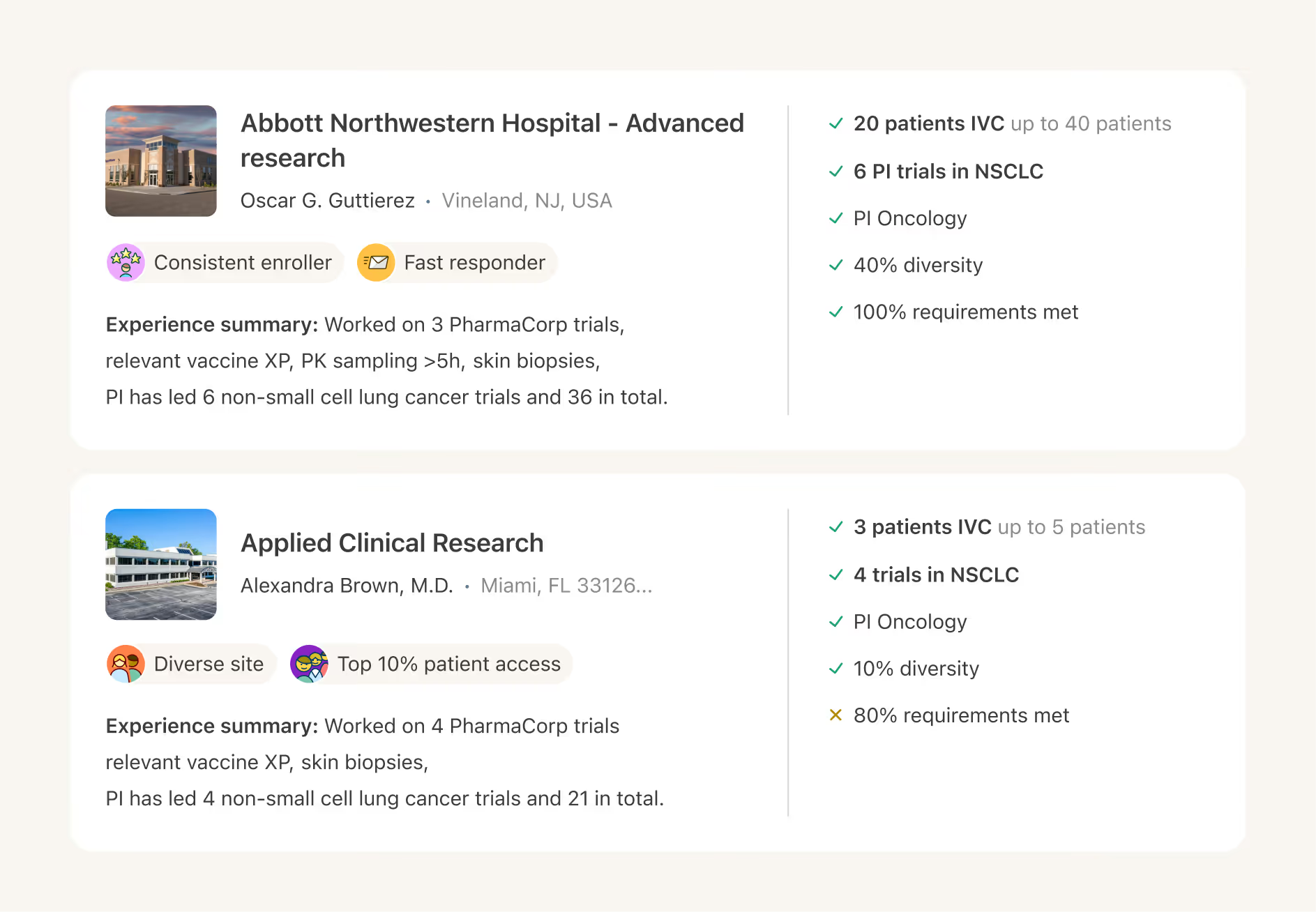
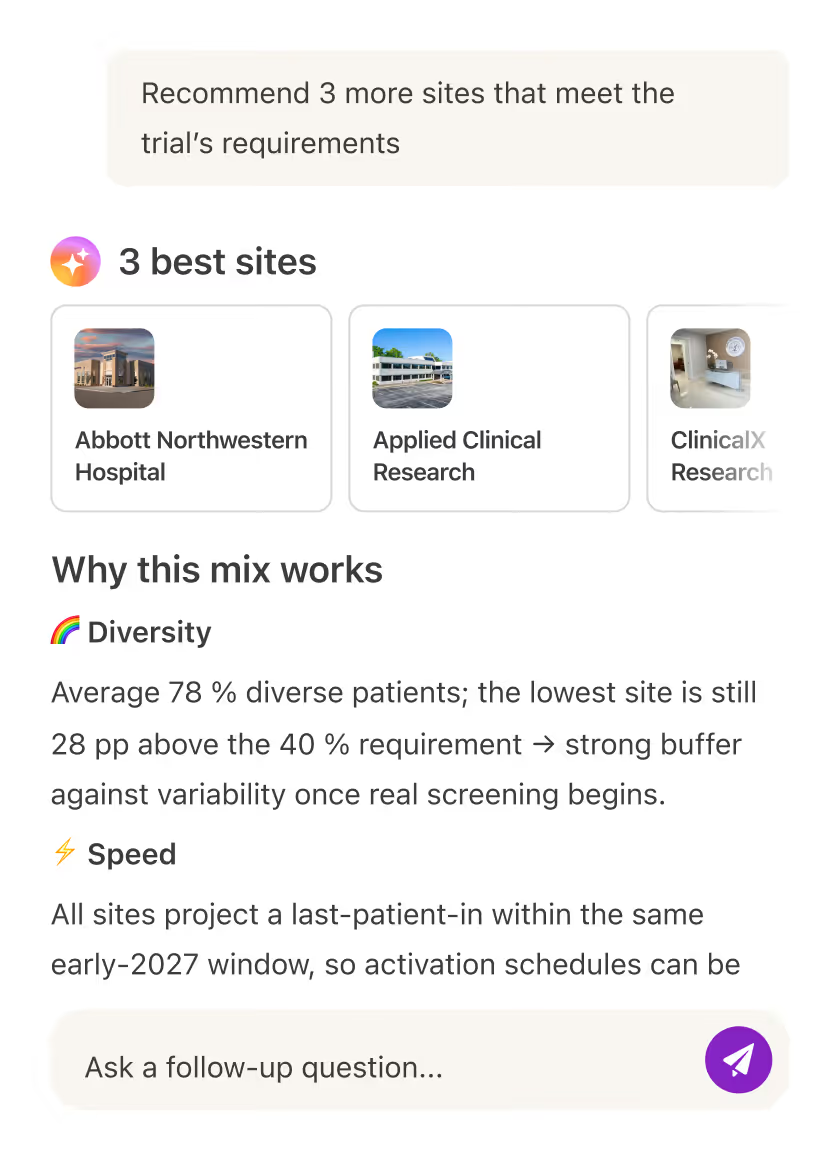
AI site selection
Confidently select the mix of sites that best aligns with your trial goals, powered by AI insights that guide better decisions.



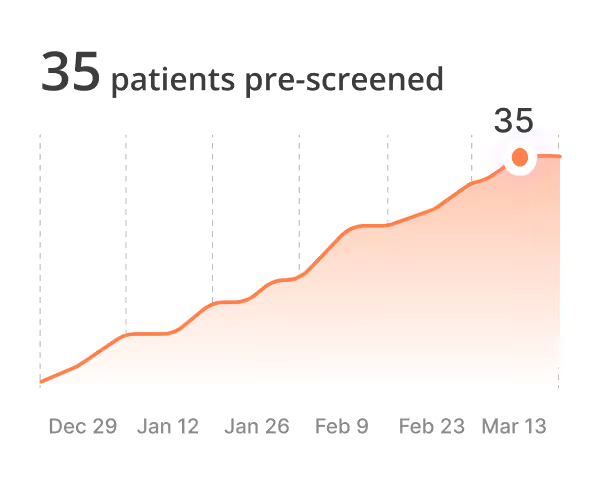
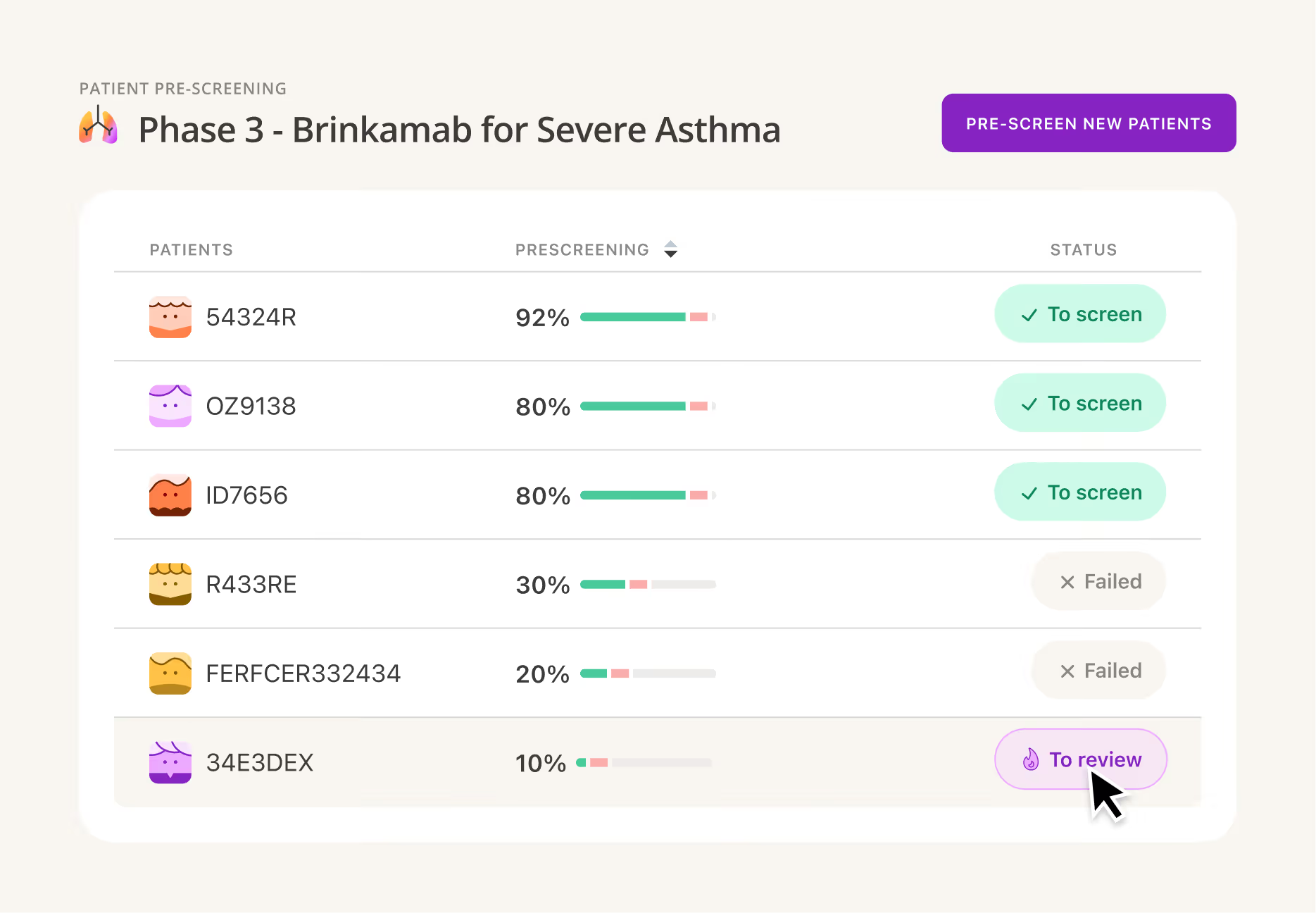
AI patient pre‑screening
Reduce site burden, accelerate chart review, and gain visibility with real-time insights by site, trial, and indication.
How it all
works together
Each step powers the next, creating a seamless flow from planning to performance. Sponsors gain early protocol insights, engage high-performing sites faster, and track real-time site performance to optimize every stage of the trial.


